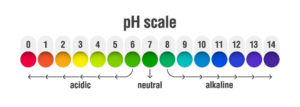
Hence at the lower end of the scale one will find the acids whereas at the higher end of the scale the bases.
The pH value of drinking water
The authorities stipulate that drinking water should have a pH of not less than 6.5 and not more than 9.5. Therefore a lot of waterworks are adding lime slurry to the raw water. To bring down the pH, to come as close as possible to a neutral pH of 7.
To make this rather dry pH value, a little bit more understanding here a few examples. The pH value of:
| acidic | neutral | alkaline |
| vinegar 2.0 | pure water 7.0 | blood 7.4 |
| sulfuric acid 2.75 | soap 10.0 | |
| acid rain 5.0 | bleach 12.5 | |
| human urine 6.0 | caustic soda 14.0 | |
| milk 6.4 |
This pH values in the table above are average values, they might vary from case to case. The pH of the human urine for instance, depends a lot on the diet, the medicines or the diseases one has. It can go as low as 5.0 considered as acidic or as high as 8.5 considered alkaline.
